
연구
Research Outcome
미래를 창조하는 포스텍 화학공학과
Stability-Controllable Self-Immobilization of Carbonic Anhydrase Fused with a Silica-Binding Tag onto Diatom Biosilica for Enzymatic CO2 Capture and Utilization
- Title of paper
- Stability-Controllable Self-Immobilization of Carbonic Anhydrase Fused with a Silica-Binding Tag onto Diatom Biosilica for Enzymatic CO2 Capture and Utilization
- Author
- [차형준교수 연구실] 바이오실리카에 고정화된 효소를 이용한 이산화탄소 저감 및 전환
- Publication in journal
- ACS Appl. Mater. Interfaces 2020, 12, 24, 27055–27063
- Publication date
- 20200617
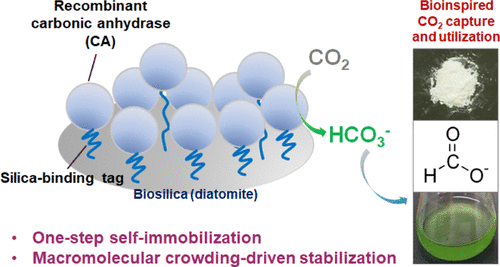
[Abstract]
Exploiting carbonic anhydrase (CA), an enzyme that catalyzes the hydration of CO2, is a powerful route for eco-friendly and cost-effective carbon capture and utilization. For successful industrial applications, the stability and reusability of CA should be improved, which necessitates enzyme immobilization. Herein, the ribosomal protein L2 (Si-tag) from Escherichia coli was utilized for the immobilization of CA onto diatom biosilica, a promising renewable support material. The Si-tag was redesigned (L2NC) and genetically fused to CA from the marine bacterium Hydrogenovibrio marinus (hmCA). One-step self-immobilization of hmCA-L2NC onto diatom biosilica by simple mixing was successfully achieved via Si-tag-mediated strong binding, showing multilayer adsorption with a maximal loading of 1.4 wt %. The immobilized enzyme showed high reusability and no enzyme leakage even under high temperature conditions. The activity of hmCA-L2NC was inversely proportional to the enzyme loading, while the stability was directly proportional to the enzyme loading. This discovered activity–stability trade-off phenomenon could be attributed to macromolecular crowding on the highly dense surface of the enzyme-immobilized biosilica. Collectively, our system not only facilitates the stability-controllable self-immobilization of enzyme via Si-tag on a diatom biosilica support for the robust, facile, and green construction of stable biocatalysts, but is also a unique model for studying the macromolecular crowding effect on surface-immobilized enzymes.
DOI: 10.1021/acsami.0c03804



